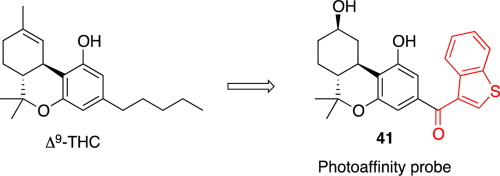
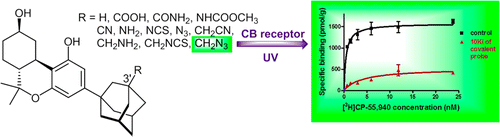
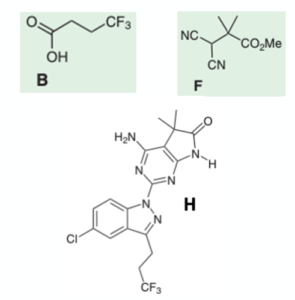
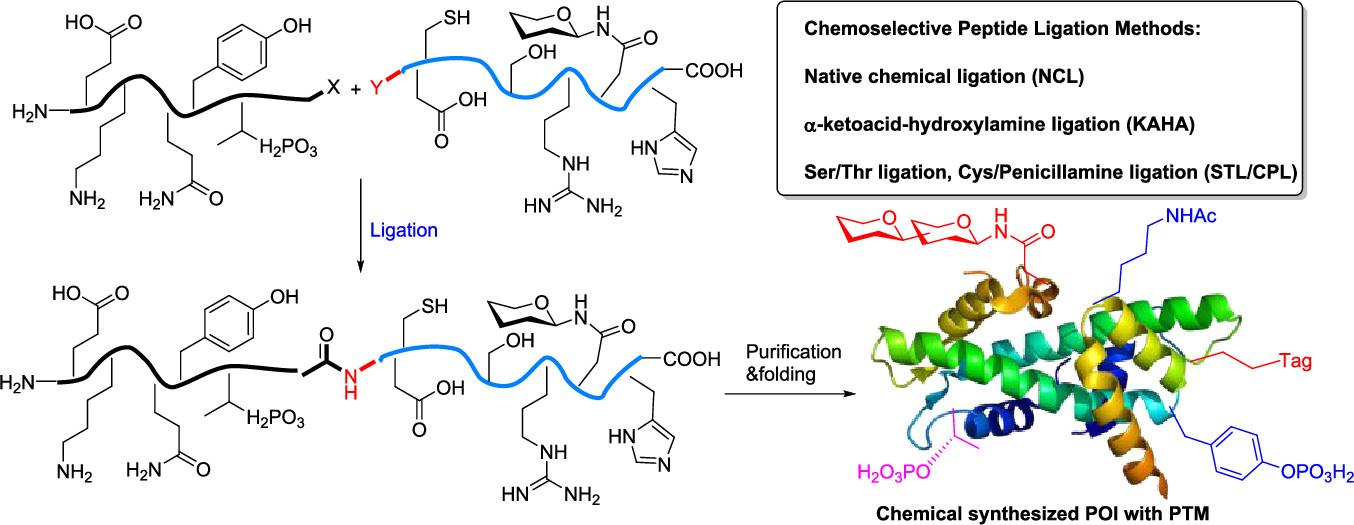
Anilinoquinazoline inhibitors of the RET kinase domain—Elaboration of the 7-position – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub.

Bioorganic & Medicinal Chemistry Letters | Recent Advances in Medicinal Chemistry | ScienceDirect.com by Elsevier
Bioorganic & Medicinal Chemistry Letters 1992: Vol 2 Index : Free Download, Borrow, and Streaming : Internet Archive
![PDF) Corrigendum to “Metal-assisted synthesis of unsymmetrical magnolol and honokiol analogs and their biological assessment as GABAA receptor ligands” [Bioorg. Med. Chem. Lett. 25/2 (2015) 400–403] PDF) Corrigendum to “Metal-assisted synthesis of unsymmetrical magnolol and honokiol analogs and their biological assessment as GABAA receptor ligands” [Bioorg. Med. Chem. Lett. 25/2 (2015) 400–403]](https://i1.rgstatic.net/publication/272568821_Corrigendum_to_Metal-assisted_synthesis_of_unsymmetrical_magnolol_and_honokiol_analogs_and_their_biological_assessment_as_GABAA_receptor_ligands_Bioorg_Med_Chem_Lett_252_2015_400-403/links/555b168208ae6fd2d8287b88/largepreview.png)
PDF) Corrigendum to “Metal-assisted synthesis of unsymmetrical magnolol and honokiol analogs and their biological assessment as GABAA receptor ligands” [Bioorg. Med. Chem. Lett. 25/2 (2015) 400–403]
Koehler MFT, Bergeron P, Blackwood EM, Bowman K, Clark KR, Firestein R, Kiefer JR, Maskos K, McCleland ML, Orren L, Salphati L,
Writing Your Next Medicinal Chemistry Article: Journal Bibliometrics and Guiding Principles for Industrial Authors

LogD Contributions of Substituents Commonly Used in Medicinal Chemistry | ACS Medicinal Chemistry Letters

PDF) Design, synthesis, and structure-activity-relationship of phenyl imidazoles as potent Smoothened antagonists